US President Donald Trump signs Right To Try bill into law

For patients who reached a dead end, the right to try lay passed by US President Donald Trump is a milestone.
With the
#RightToTry Law I signed today, patients with life threatening illnesses will finally have access to experimental treatments that could improve or even cure their conditions. These are experimental treatments and products that have shown great promise.- US President Donald Trump
See details below compiled from the Internet
RIGHT TO TRY: Congress passed the Right to Try Act of 2017, sending a priority bill to President Donald J. Trump for his signature.
- The “Trickett Wendler, Frank Mongiello, Jordan McLinn, and Matthew Belllina Right to Try Act of 2017” passed Congress on May 22, 2018.
- The bill amends Federal law to allow certain unapproved, experimental drugs to be administered to terminally ill patients who have exhausted all approved treatment options and are unable to participate in clinical drug trials.
- Eligible drugs must have undergone the Food and Drug Administration’s (FDA) Phase I (safety) testing.
- The bill requires any manufacturer or sponsor of an eligible investigational drug to report to the FDA on any use of the drug on a “Right to Try” basis.
- The FDA will post an annual summary report of “Right to Try” use on its website.
- The bill limits the liability of drug sponsors, manufacturers, prescribers, or dispensers that provide or decline to provide an eligible investigational drug to an eligible patient.
A RIGHT TO LIFE: Right to try legislation returns treatment decisions back to patients, giving them the right to make healthcare choices that could save their lives.
- “Right to Try” gives the over 1 million Americans who die from a terminal illness every year a new tool to fight and make potentially lifesaving decisions about their treatment.
- According to the National Cancer Institute, an estimated 609,640 Americans will die from cancer alone in 2018.
- Prior to this legislation, options for terminally patients who had exhausted all treatment options were extremely limited:
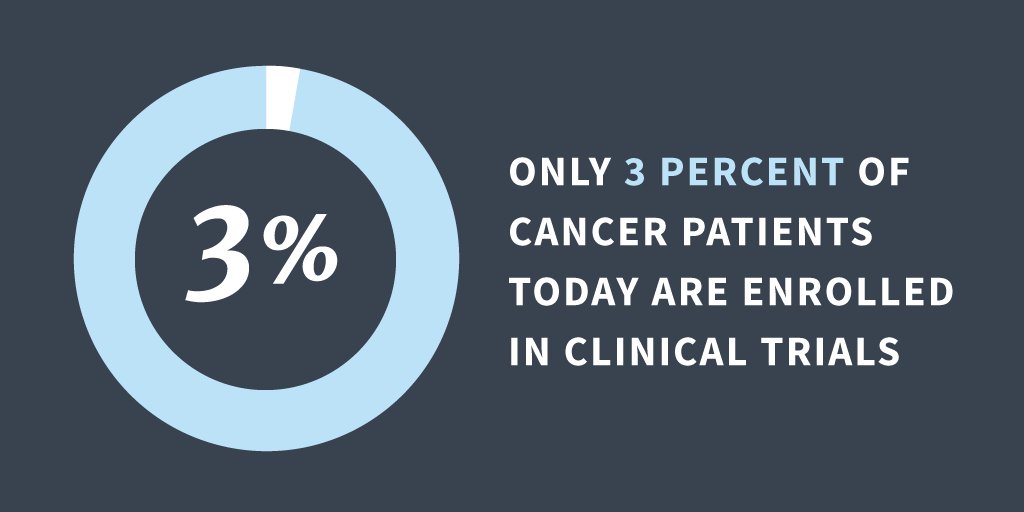
- Many terminally ill patients were considered too sick for clinical trials of new medication. In fact, only 3 percent of cancer patients today are enrolled in clinical trials.
- The FDA’s “compassionate use” application is complicated and time-consuming, resulting in only about 1,200 approved applications per year.
- With President Trump’s signature the Federal government joins with 40 States that have approved the right of terminally ill patients to try potentially lifesaving drugs.